Hydrogen atomic number of neutrons2/12/2024 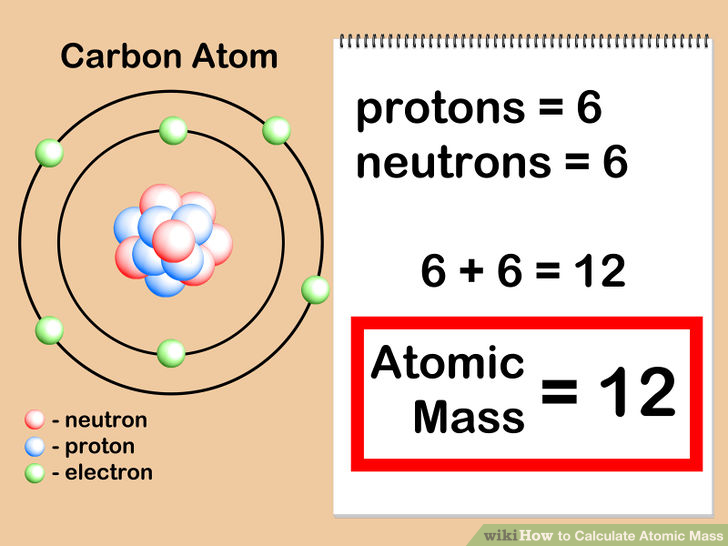
Chemical properties include the physical state of the element (gas, liquid, or solid), the types of bonds the element can form, how it reacts with other elements, etc. We can define an atom as the simplest particle of an element that has the chemical properties of that element. To determine the number of neutrons in this isotope, we subtract 26 from 56: 56 − 26 = 30, so there are 30 neutrons in this atom.Inorganic Chemistry (Atomic Structure) AtomsĪll elements are composed of extremely small particles of matter called atoms. The 26 is the atomic number (which is the same for all iron atoms), while the 56 is the mass number of the isotope. Which indicates a particular isotope of iron. Thus, we might see 26 56 atomic number → mass number → Fe Atomic numbers are often listed as a subscript on the left side of an element’s symbol. Given the mass number for a nucleus (and knowing the atomic number of that particular atom), you can determine the number of neutrons by subtracting the atomic number from the mass number.Ī simple way of indicating the mass number of a particular isotope is to list it as a superscript on the left side of an element’s symbol. of an atom is the sum of the numbers of protons and neutrons in the nucleus. The mass number The sum of the numbers of protons and neutrons in a nucleus of an atom. 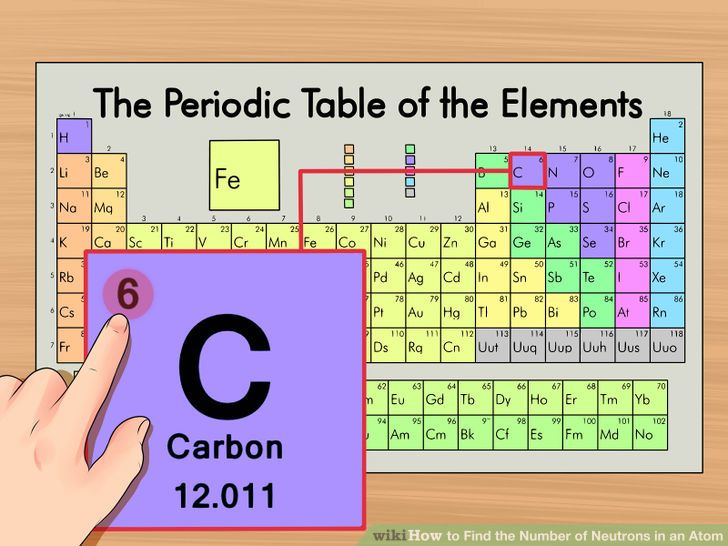
When scientists discuss individual isotopes, they need an efficient way to specify the number of neutrons in any particular nucleus. In fact, there are currently over 3,500 isotopes known for all the elements. Most elements exist as mixtures of isotopes. 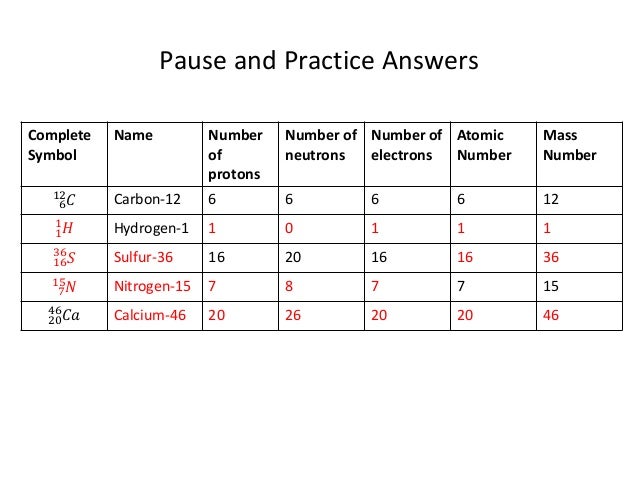
Dalton thought that all atoms of the same element were exactly the same. The discovery of isotopes required a minor change in Dalton’s atomic theory. Figure 2.6 "Isotopes of Hydrogen" compares the three isotopes of hydrogen. 
An extremely rare hydrogen isotope, tritium, has 1 proton and 2 neutrons in its nucleus. About 1 in 10,000 hydrogen nuclei, however, also has a neutron this particular isotope is called deuterium. Most hydrogen atoms have a nucleus with only a single proton. Naturally occurring carbon on Earth, therefore, is actually a mixture of isotopes, albeit a mixture that is 99% carbon with 6 neutrons in each nucleus.Īn important series of isotopes is found with hydrogen atoms. For example, 99% of the carbon atoms on Earth have 6 neutrons and 6 protons in their nuclei about 1% of the carbon atoms have 7 neutrons in their nuclei. Atoms of the same element that have different numbers of neutrons are called isotopes Atoms of the same element that have different numbers of neutrons. However, it was found that atoms of the same element can have different numbers of neutrons. How many neutrons are in atoms of a particular element? At first it was thought that the number of neutrons in a nucleus was also characteristic of an element. The largest atoms have over 100 protons in their nuclei. From this table, you can determine the number of protons in the nucleus of any element. Chapter 21 "Appendix: Periodic Table of the Elements" lists the elements and their atomic numbers. There is no such thing as a hydrogen atom with 2 protons in its nucleus a nucleus with 2 protons would be a helium atom. Helium has the atomic number 2 all helium atoms have 2 protons in their nuclei. This discovery was so important to our understanding of atoms that the number of protons in the nucleus of an atom is called the atomic number The number of protons in the nucleus of an atom.įor example, hydrogen has the atomic number 1 all hydrogen atoms have 1 proton in their nuclei. Furthermore, different elements have a different number of protons in their nuclei, so the number of protons in the nucleus of an atom is characteristic of a particular element. In other words, all atoms of the same element have the same number of protons. In the 1910s, experiments with X rays led to this useful conclusion: the magnitude of the positive charge in the nucleus of every atom of a particular element is the same.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
